Summary
MIT researchers have unveiled an idea that sounds futuristic but is aimed at a very current medical crisis: injectable “mini livers” designed to help patients whose own livers are failing. The technology is meant for people who are waiting for a transplant, as well as many who may never qualify for one because they are too sick to undergo major surgery. MIT says more than 10,000 Americans with chronic liver disease are on the transplant waiting list, while donated organs remain far too scarce to meet demand.
The new approach does not try to replace the damaged liver outright. Instead, the MIT team describes the grafts as “satellite livers” — small engineered tissue implants that could be injected into the body and take over part of the liver’s workload while the original organ remains in place. In tests in mice, the researchers found that these injected liver cells stayed viable for at least eight weeks and continued making enzymes and proteins normally produced by the liver.
Don't miss anything!
Join our community:
“We think of these as satellite livers. If we could deliver these cells into the body, while leaving the sick organ in place, that would provide booster function,” says Sangeeta Bhatia, the John and Dorothy Wilson Professor of Health Sciences and Technology and of Electrical Engineering and Computer Science at MIT, and a member of MIT’s Koch Institute for Integrative Cancer Research and the Institute for Medical Engineering and Science (IMES).
Bhatia is the senior author of the new study, which appears today in the journal Cell Biomaterials. MIT postdoc Vardhman Kumar is the paper’s lead author.
That distinction matters. A traditional liver transplant is not only limited by the number of donor organs; it is also a major surgical event that many fragile patients cannot tolerate. MIT’s concept is built around a simpler goal: restoring enough liver function to stabilize the patient, reduce risk, and potentially buy time. The team says the technology could work either as an alternative to surgery in some cases or as a bridge to transplantation until a donor organ becomes available.
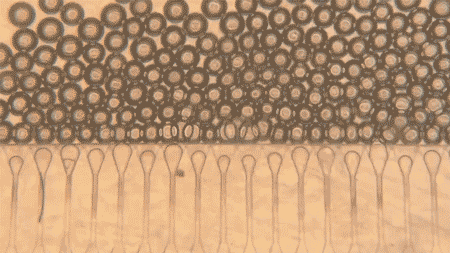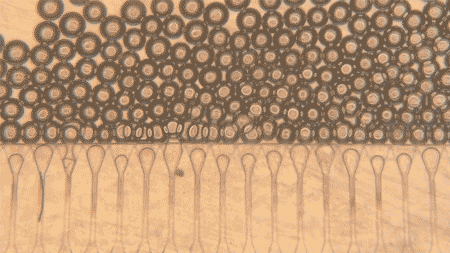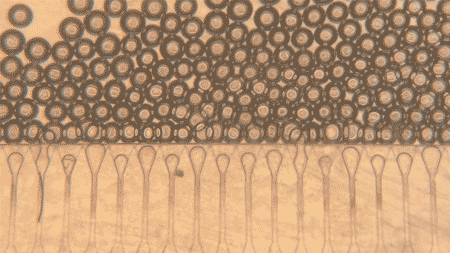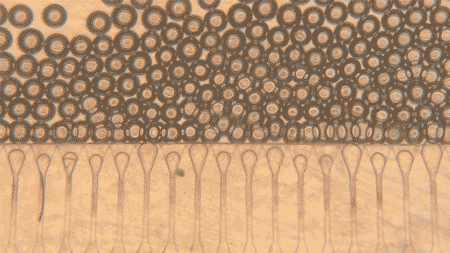
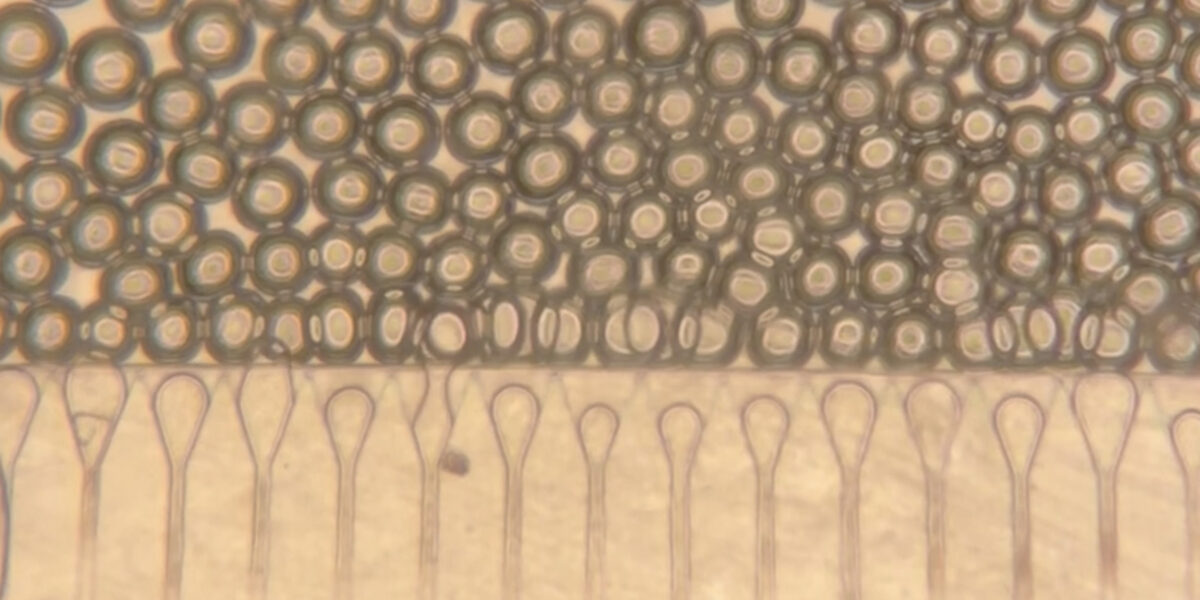
At the heart of the system are hepatocytes, the cells that perform much of the liver’s work. But injecting those cells alone is usually not enough, because they tend not to organize well or connect efficiently with the body after delivery. MIT’s answer was to pair the cells with hydrogel microspheres, tiny engineered particles that help create a supportive niche inside the body. The spheres can be injected through a syringe while packed closely together, then regain their structure once inside, helping the liver cells remain localized and link up with nearby blood vessels.
The researchers also added fibroblast cells, which help support the hepatocytes and encourage blood vessel growth into the graft. That blood supply is the key to survival: the more quickly the implanted cells connect to the host circulation, the better their chances of functioning like real liver tissue. According to MIT, that is exactly what happened in the mouse experiments, where new blood vessels formed close to the implanted hepatocytes and helped keep them healthy.
Another reason the work stands out is that the grafts were delivered without open surgery. The team developed an ultrasound-guided injection method, allowing them to place the cell mixture precisely and then monitor the implant later using ultrasound as well. In this study, the mini livers were injected into fat tissue in the abdomen, but the researchers say future versions could potentially be delivered to other sites, such as near the kidneys or into the spleen, as long as there is enough space and access to blood vessels.
That flexibility could make the concept far more practical than earlier tissue-engineering strategies that depended on surgically implanted scaffolds. MIT says one of the biggest advantages here is that if a patient eventually needed more grafts or another round of therapy, the barriers would be much lower than repeating another major operation.
Still, this is early-stage science, and the researchers are not presenting it as a ready-made clinical treatment. The current version would likely require patients to take immunosuppressive drugs, because the implanted cells could still be attacked by the immune system. The team is now exploring whether they can develop more “stealthy” liver cells that avoid immune detection, or use the hydrogel system itself to deliver immunosuppressive drugs locally.
The broader medical need is obvious. The liver is involved in roughly 500 essential functions, including blood clotting, clearing bacteria from the bloodstream, and metabolizing drugs. When it starts to fail, the damage can quickly become systemic. That is why a therapy that can restore even part of that function — without immediately requiring a transplant — would represent a major shift in care.
For now, the MIT breakthrough is best understood as a promising proof of concept rather than a near-term cure. But it is also more than a laboratory curiosity. It offers a credible glimpse of a future in which doctors may be able to support a failing liver not by replacing it at once, but by injecting a second, smaller source of liver function into the body and keeping patients alive long enough for the next step in treatment.